|
Water "creates" an equilibrium based on solute concentration inside and outside of your cells.Īll of that is well and good, but how do we actually use fluids for therapy.įor starters, IV fluids are broken down into two major branches: Crystalloids and Colloids. And it does this based on the rules governed by oncotic pressure. The lipid bi-layer cell membrane does a pretty good impression of the semi-permeable membrane in the image above. Most solutes (Na, K, Ca, Mg, etc.) cannot freely pass into or out of your cells. Like the membraned beaker of 7th grade bio, so are the cells of your body. Remember in 7th grade biology class when you learned about diffusion and osmosis? How an equilibrium is naturally created as water diffuses from a low concentration of solute to a high concentration? This 60/40 split of intra/extracellular fluid is created and driven by oncotic pressure. This all can be easily demonstrated with some flowcharts: If you're struggling to visualize this (you're not the only one), just think of it as everything that is not inside of a cell or in the blood stream. It lives in-between and surrounding your cells. What about the remaining 75% of the extracellular fluid? This fluid is called interstitial fluid.What the crap is that? It's fluid that is said to "bathe" your cells. It's what trauma victims "bleed out." In total, it's roughly about 3 liters. It supplies oxygen and nutrients to the body. Intravascular (IV) fluid is what most people think of when they say "body fluid." It's the blood in your arteries and veins. Surprisingly, only 25% of this fluid is intravascular fluid. The 40% that is our extracellular fluid, is broken down a little further. The remaining 40% is outside of cells (extracellular). About 60% of total body fluid lives inside of cells (intracellular). Let's start with a breakdown of fluid distribution in the body. 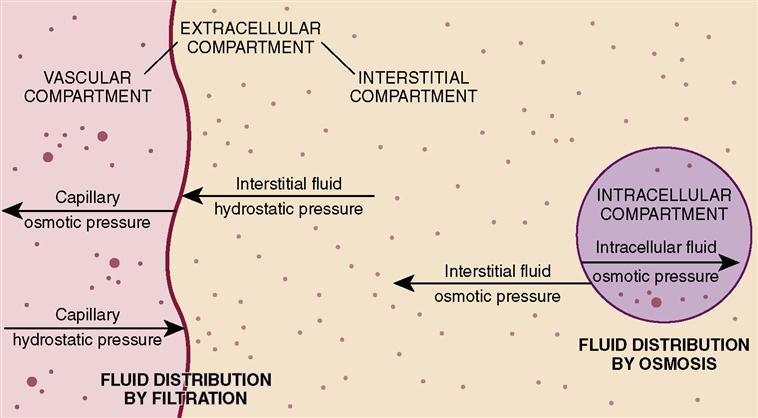
If you’re the note-taking type, you can get a downloadable and printer-friendly copy of this article, you can get that here. Typically, you will use either D5W or sterile water if you're adding that much bicarb.īTW, we’re about to get real. So although fluid management may not be the "sexiest" topic for pharmacists you'll be doing yourself (and your patients) a favor by having an understanding of what's going on in the wonderful world of fluids.Īnd, if the suspense is killing you: Putting 3 amps of bicarb in NS creates a hypertonic solution. I went on to see that fluid management is one of the most common interventions encountered in a hospital system. Even in retail (ahem: community) pharmacy, there are opportunities to make a difference in the fluid status of your CHF, liver failure, and pregnant patients. It was in that moment that I perfected the classic "I'm not sure, but let me look it up really quick" routine. I have no idea what 3 amps of bicarb can go into. I don't remember really talking about fluids at all during pharmacy school. Do I just order that to go in normal saline?" "We want to give her 150 mEq of bicarb per liter until she gets transferred. This will alkalinize the urine, which will help the patient clear the ridiculous dose of methotrexate (about 17 GRAMS) she'll be getting later. While waiting for a bed, the resident explains that we are going to pre-hydrate her with bicarbonate. She has a primary CNS lymphoma and they are treating with high dose methotrexate. Rounds begin, and I learn that we have a patient that will be transferred to the chemo floor later that day. And I woke up hours before that because I hadn't yet lost my motivation for exercise and general health and wellness. 
Rounds start at 9am, but I arrived hours before to work up my patients. It's still early in the residency, and I'm already tired. I'm on my first rotation, covering is an IMC unit (which has patients less sick than an ICU, but more sick than a regular medicine floor). I'm still a little nervous with the whole "residency" thing.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
